Biomass can be converted into energy (heat or electricity) or energy carriers (charcoal, oil, or gas) using both thermochemical and biochemical conversion technologies. Combustion is the most developed and most frequently applied process because of its low costs and high reliability.
However, combustion technologies deserve continuous attention from developers in order to remain competitive with the other options.
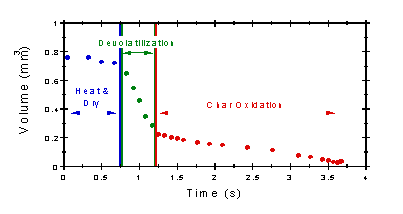 |
| Distinct stages in the process of combustion of a particle: (1) heating and drying, (2) devolatilization and (3) char oxidation |
During combustion, the biomass first loses its moisture at temperatures up to 100°C, using heat from other particles that release their heat value. As the dried particle heats up, volatile gases containing hydrocarbons, CO, CH4 and other gaseous components are released. In a combustion process, these gases contribute about 70% of the heating value of the biomass. Finally, char oxidises and ash remains.
 |
| Wood chip combustion on a grate furnace. Fuel enters the furnace at the right hand side and devolatilizes as it is transported to the left. At the left hand side, remaining char burns out. |
The combustion installation needs to be properly designed for a specific fuel type in order to guarantee adequate combustion quality and low emissions. Emissions caused by incomplete combustion are usually a result of either:
- poor mixing of combustion air and fuel in the combustion chamber, giving local fuel-rich combustion zones
- an overall lack of available oxygen
- combustion temperatures that are too low
- residence times that are too short
- radical concentrations that are too low
Through experiments and modelling, new boiler geometries and combustion concepts have been developed that result in significantly lower emissions.
Examples of such developments are reburning of fuel, air staging, air preheating, radiation shields, advanced combustion control systems, application of novel materials, etc. Task 32 aims to be instrumental in


